Cobatt Oxide Co3o4 Powder

Kaoaohai
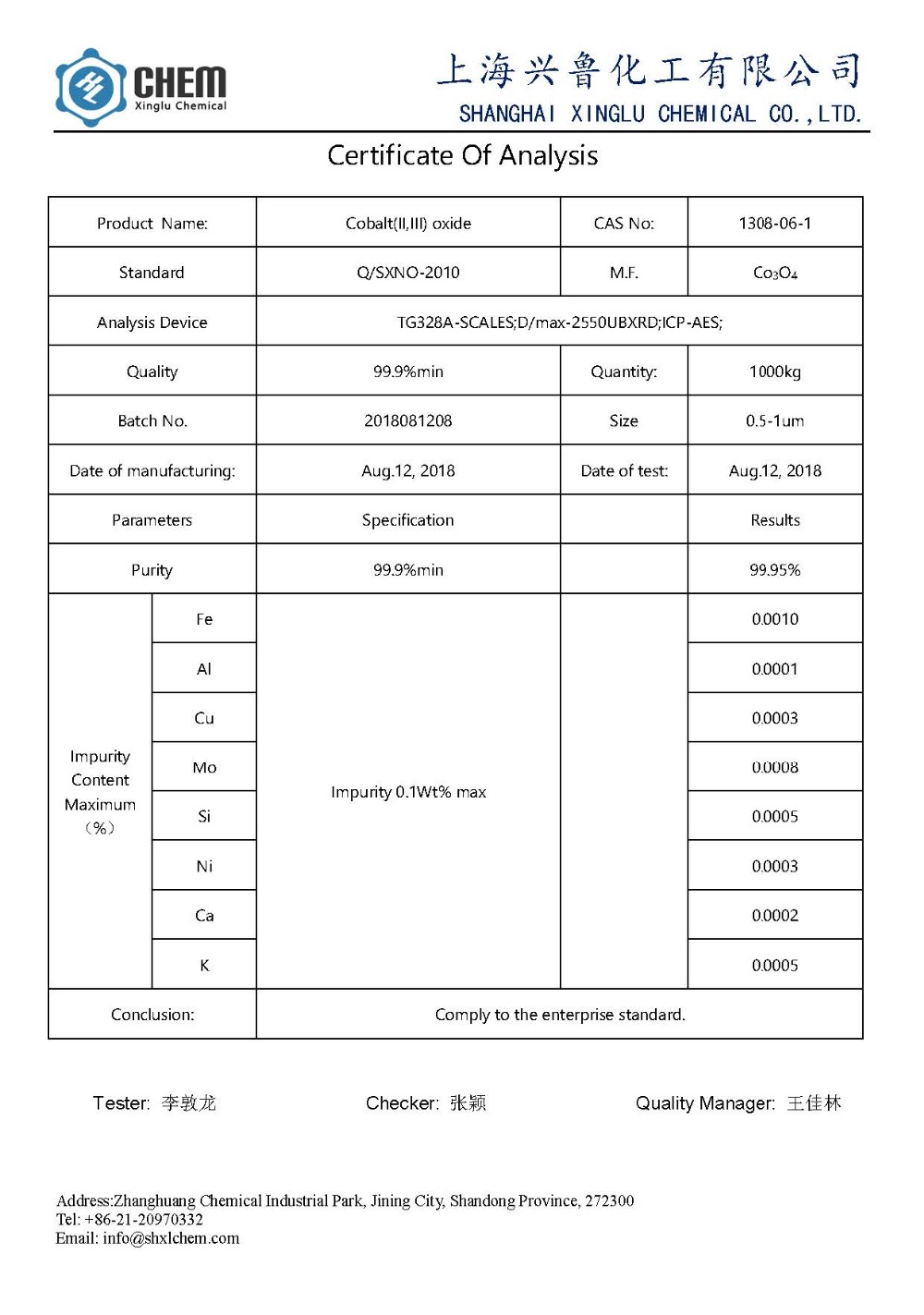
1. Makey: NanoCoplett oxideCo3o4 powder
2.Pugh: 99.9% min
3.Appearacne: hina hina hina
4.Partticle nui: 50nm
5.ssa: 30-80 m2 / g
Waiwai:
Ka hoʻolahaʻana i ka ea, maʻalahi e hoʻohui i ka waiu, akāʻaʻole e hoʻokomo i nā ulu wai wai. He meaʻala i ka nitric acid. I ka wa i hoʻomoʻia ma luna o 1200 oc, e wāwahiʻiaʻo Nano-cubalat oxde oxde i loko o ka obide sub-cub-cusdet cub-cuside sub-cuside sub-cuside sub-cuside sub-cuside sub-cub-cuside sub-cuside sub-cuside. I loko o ka kīwī hydrogen,ʻo Nano-cubalt i hoʻomehanaʻiaʻo Oxide i 900 oc, e hoʻololiʻia i loko o ketallelo metalt. Cobalt (II, III)ʻO OMID ka coctaical coctaile me keʻano cocopiad me keʻano heʻano cor3o4. He'eleʻeleʻeleʻele, a me kahi hui pūʻana me ka hoʻopiliʻana, a me nā huiʻelua a me nā coli). Hiki ke hoʻokumuʻia e like me ke coiicoiii2o4 a iʻole cou.co2o3. Cobalt (II) Oxide, COOL, e hoʻohuli i ka CO3O4 inā hertated a puni 600-700 ° C i ka lewa. Ma luna o 900 ° C, CO CO CONTAT.
Ke noi:
ʻO Catlysis, SuperCondactor, nā ceramtics a me nāʻano'ē aʻe e like me nā mea nui loa; E like me ka mea lawe catalyst a me ka mea lawe catalyst a me ka mea electrode ikaika; No ke aniani, nā kāwele porcelain a me nā puaʻa; ʻO Lupa Pūnaehana Kūʻai Kūʻai a me kahi catalyst no ka synthes organic; Nā gensior geggle a me nā mea lako'ē aʻe; Kālai; O ke aniani a me nā mea like; No ka kakai limahana semiconductor, uila chemesics, lithium ion Nā mea hana uila; Eamia; Nā huila; Heterogeneous No laila e lele i nā meaʻike o ka ikehu.
HAAKAANA:

He aha kā mākou e hāʻawi ai: